Aqueous zinc–halogen batteries have attracted extensive attention for next-generation energy storage because of their high safety, low cost, and relatively high energy density. They show broad application prospects in wearable electronics, smart healthcare, portable devices, new-energy transportation, and aerospace. However, these batteries have long been limited by several key challenges, including the dissolution of active halogen species, severe shuttle effects, and sluggish interfacial reaction kinetics, which lead to rapid capacity fading, insufficient rate capability, and limited cycling stability. At the same time, conventional cathode host materials often struggle to simultaneously provide fast electron transport, strong halogen anchoring, structural stability, and mechanical flexibility, thereby restricting the development of high-performance aqueous zinc–halogen batteries.
To address these issues, the team led by Prof. Peixin Zhang, Assistant Prof. Yaokang Zhang, and Prof. Xuechang Zhou at Shenzhen University, in collaboration with Associate Prof. Chi Zhang from Sun Yat-sen University, developed a Trinity Cooperative Electrode (TCE) strategy. This electrode integrates the conductive polymer PEDOT:PSS, a tannic acid/β-cyclodextrin (TA/β-CD) supramolecular matrix, and a crosslinked poly(vinyl alcohol) (PVA) elastic network into one unified architecture. Through this design, the electrode simultaneously achieves enhanced electronic conduction, halogen immobilization, interfacial electrocatalysis, and mechanical robustness. The strategy significantly improves the rate capability, cycling stability, and environmental adaptability of aqueous zinc–halogen batteries, and is further extended to multi-halogen systems and flexible energy-storage devices, offering a new route for the design of high-energy, long-life cathode materials.

Recently, the related work was published in Angewandte Chemie International Edition under the title “Trinity Cooperative Electrode as a High-Performance Electrocatalytic Host for Ultra-Stable, High-Rate Zinc–Halogen Batteries.” The co-first authors are Mengnan Lai and Ming Yang, both PhD students at Shenzhen University. The corresponding authors are Associate Prof. Chi Zhang of Sun Yat-sen University, and Prof. Peixin Zhang, Assistant Prof. Yaokang Zhang, and Prof. Xuechang Zhou of Shenzhen University.

Figure 1. Schematic illustration of the design concept and functional architecture of the Trinity Cooperative Electrode (TCE)
Building on their previous research in conductive polymer composites and supramolecular systems, the team successfully constructed a TCE cathode material system with both structural stability and electrochemical activity. Using a simple solution-based process, a PEDOT:PSS dispersion, a PVA solution containing glutaraldehyde crosslinker, and a TA/β-CD supramolecular matrix were uniformly mixed. Through chemical crosslinking and hydrogen-bond interactions, a robust three-dimensional composite network was formed. The team then systematically characterized the chemical composition, microstructure, conductivity, mechanical flexibility, and interfacial stability of TCE using FTIR, XRD, Raman spectroscopy, AFM, SEM, TEM, tensile tests, and peeling tests. Electrochemical measurements, including cyclic voltammetry, galvanostatic charge–discharge, in situ FTIR, in situ Raman, XPS, EIS/DRT, and GITT, were further carried out to clarify how TCE promotes reversible halogen conversion and fast interfacial kinetics. Finally, TCE was extended to Zn–Br2 and Zn–I/Br multi-halogen systems, and flexible pouch cells were assembled to verify its application potential under low-temperature and wearable conditions.

Figure 2. Structural characterization of TCE and its halogen affinity
A series of characterization results confirmed the successful construction of the TCE composite electrode and its excellent overall performance. XRD and Raman analyses showed that the π–π interaction between TA and PEDOT promoted the conformational transition of PEDOT:PSS from the benzoid structure to the more conductive quinoid structure, thereby significantly improving electron transport. The electrical conductivity increased dramatically from 0.5 S cm⁻1 for pristine PEDOT:PSS to 708 S cm⁻1 for TCE. Meanwhile, the crosslinked PVA network endowed the material with good mechanical properties. TCE exhibited a tensile stress of up to 2 MPa and a breaking strain of about 50%, while maintaining structural integrity even after peeling tests, demonstrating excellent adhesion and cohesive stability.
In terms of halogen adsorption, Raman, UV–vis, and EDS results showed that TCE had a strong ability to immobilize iodine species, with almost no obvious iodine loss after immersion in a ZnI₂ solution. Its water contact angle was only 51°, much lower than that of activated carbon and pristine PEDOT:PSS, indicating better wettability and electrolyte infiltration. Theoretical calculations further revealed that PEDOT, PVA, β-CD, and TA all provided thermodynamically favorable adsorption sites for iodine species. Among them, PEDOT exhibited adsorption energies of −1.29 eV for I2 and −2.48 eV for I3⁻, significantly stronger than those of conventional carbon materials. This strong affinity lays the foundation for suppressing shuttle effects and improving reaction reversibility.

Figure 3. Electrochemical performance of TCE-based Zn–I2 batteries
Benefiting from its trinity cooperative structure, TCE delivered excellent energy-storage performance in aqueous zinc–iodine batteries. Compared with activated carbon and pristine PEDOT:PSS, the TCE electrode showed a larger CV integral area and lower polarization. Its Tafel slope during oxidation was only 42 mV dec⁻1, indicating faster electrocatalytic reaction kinetics.
In ZnI2 + Zn(CF3SO3)2 electrolyte, TCE-based batteries delivered a stable reversible capacity of 255 mAh g⁻1 at 1 A g⁻1 and remained stable for more than 2700 cycles. Even at the ultrahigh rate of 50 A g⁻1, the battery still maintained a capacity above 150 mAh g⁻1 for over 50,000 cycles, with a capacity retention of more than 92%. In terms of rate capability, the initial capacity reached 338.31 mAh g⁻1 at 0.5 A g⁻1, and a high capacity of 175.83 mAh g⁻1 was still retained at 50 A g⁻1, demonstrating remarkable adaptability across a 100-fold change in current density. The maximum energy density reached 348 Wh kg⁻1, and even at an ultrahigh power density of 510,000 W kg⁻1, the system still maintained an energy density of 191 Wh kg⁻1, outperforming many previously reported Zn–I2 cathode systems.
Notably, TCE also showed excellent low-temperature performance. At −10 °C, it still delivered a stable discharge capacity of about 230 mAh g⁻1 at 1 A g⁻1 and maintained stable cycling over 1000 cycles, demonstrating strong environmental adaptability.

Figure 4. In situ/ex situ mechanistic characterization of TCE during charge–discharge
To further understand the energy-storage mechanism, the team combined in situ FTIR, in situ Raman, and ex situ XPS analyses. The results showed that iodine storage in TCE mainly involved the reversible conversion among I⁻, I3⁻, and I5⁻. During charging, Raman peaks associated with I3⁻ and I5⁻ became more prominent; during discharging, these peaks gradually weakened, indicating the reversible participation of halogen intermediates in the electrochemical process. At the same time, the vibrational peaks of the PEDOT backbone shifted reversibly, suggesting that the conductive polymer framework underwent dynamic doping during halogen conversion and actively participated in interfacial charge transfer.
XPS results further confirmed that the chemical environment of sulfur in PEDOT and PSS changed reversibly during cycling, indicating that TCE is not merely a passive host, but an active electrocatalytic interface. SEM and EDS analyses also showed that Zn and I were uniformly distributed on the electrode surface in the charged state and were greatly reduced after discharge, confirming highly efficient and reversible interfacial electrochemical reactions.

Figure 5. Reaction kinetics analysis of TCE
Kinetic studies showed that the high-rate performance of TCE originated from its pronounced pseudocapacitive behavior and fast ion diffusion capability. CV analysis indicated that the peak current of TCE at different scan rates followed a high b-value relationship, suggesting that its energy-storage process was mainly governed by surface-controlled reactions. Further quantitative analysis showed that within the scan-rate range of 0.1–1.0 mV s⁻1, the capacitive contribution increased from 83.12% to 94.17%, significantly higher than that of the control electrodes.
GITT results showed that the Zn2+ diffusion coefficient in TCE reached 2.7 × 10⁻9 to 4.89 × 10⁻10 cm2 s⁻1, much higher than that of pristine PEDOT:PSS. In situ EIS and DRT analyses further demonstrated that TCE maintained low impedance throughout the charge–discharge process, with highly efficient and stable interfacial charge-transfer behavior. These results indicate that the trinity cooperative structure creates an efficient interfacial reaction environment through strong halogen adsorption, fast electron transport, and smooth ion migration.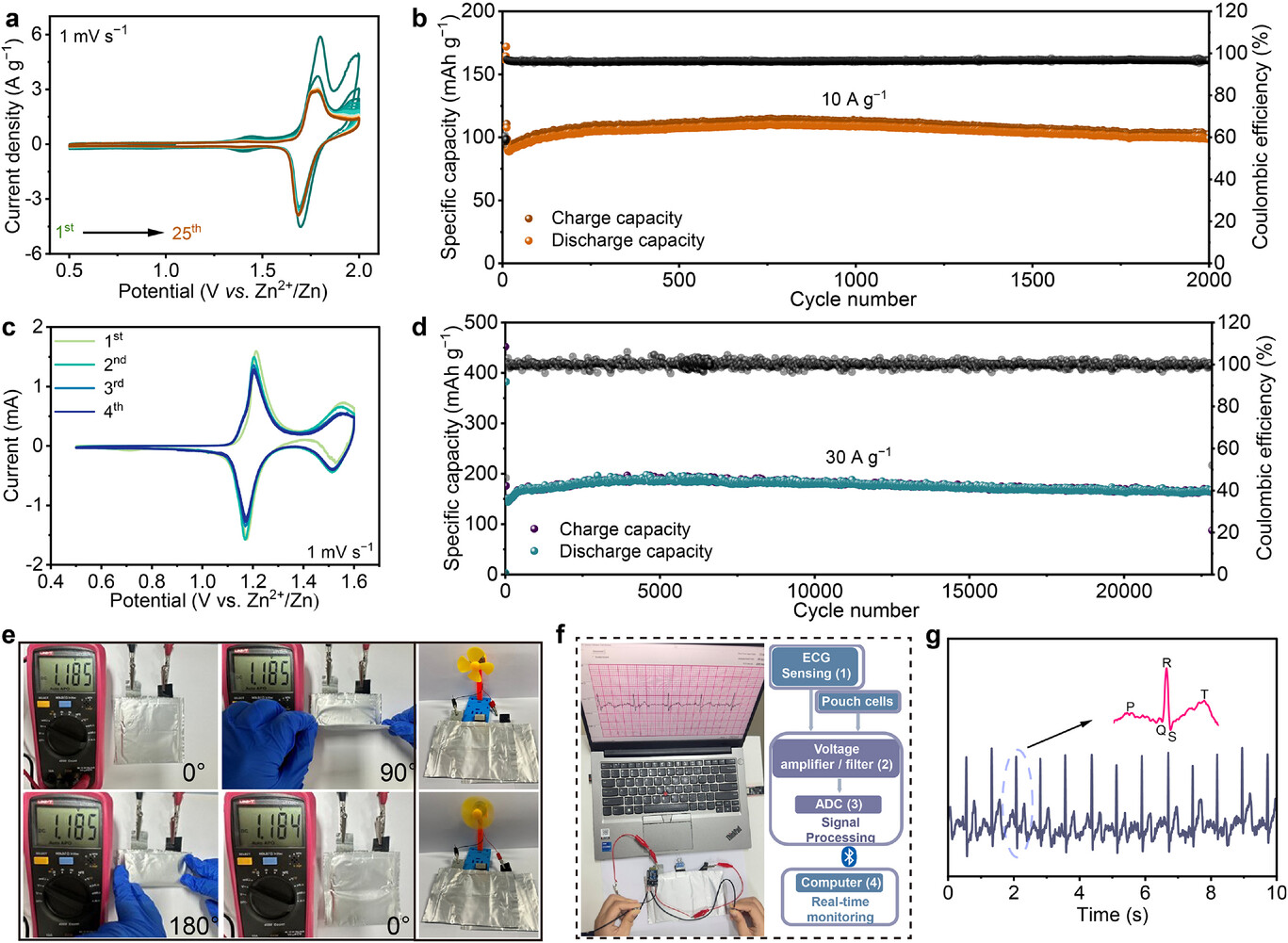
Figure 6. Multi-halogen systems and flexible pouch-cell applications
Beyond the Zn–I2 system, TCE can also be extended to other aqueous zinc–halogen batteries. In Zn–Br2 batteries, TCE maintained a stable capacity of about 100 mAh g⁻1 after 2000 cycles at 10 A g⁻1, with a Coulombic efficiency close to 100%. Furthermore, in a ternary multi-halogen electrolyte of Zn(CF3SO3)2 + ZnI2 + ZnBr2, TCE induced the formation of a stable IBr₂⁻ intermediate, activating a sequential multielectron redox pathway and thereby significantly improving the capacity and stability of the system. Under these conditions, the battery maintained a specific capacity of 165 mAh g⁻1 at 30 A g⁻1 with 87% capacity retention after 22,000 cycles.
Flexible pouch cells based on TCE also exhibited stable output voltages under bending angles of 0°, 90°, and 180°, demonstrating excellent mechanical reliability. The team further used the pouch cell to power a wireless electrocardiogram (ECG) monitoring system, successfully achieving real-time signal acquisition and display. This result highlights the promise of TCE-based batteries for flexible electronics and wearable healthcare devices.
This study proposes a trinity cooperative electrode design strategy that organically integrates a conductive polymer, a supramolecular matrix, and an elastic polymer network. By doing so, it effectively addresses key challenges in aqueous zinc–halogen batteries, including halogen shuttle, sluggish interfacial kinetics, and insufficient structural stability, enabling simultaneous breakthroughs in high capacity, ultralong cycle life, high-rate operation, and flexible-device applications. The work provides new theoretical insight and a practical design route for next-generation high-energy, ultra-stable aqueous zinc–halogen battery cathodes, while also opening new opportunities for wearable energy-storage devices.
This research was supported by the National Natural Science Foundation of China, the Shenzhen Science and Technology Program, the Scientific Foundation for Youth Scholars of Shenzhen University, and the Research Team Cultivation Program of Shenzhen University.
Paper Information
Mengnan Lai#, Ming Yang#, Chi Zhang*, Maryam A. Rafiei, Shuning Li, Chuan Xie, Guangliang Liu, Peixin Zhang*, Yaokang Zhang*, Xuechang Zhou*. Trinity Cooperative Electrode as a High-Performance Electrocatalytic Host for Ultra-Stable, High-Rate Zinc–Halogen Batteries. Angewandte Chemie International Edition, 2026, e3832443. DOI: 10.1002/anie.3832443
