Professor Zhang Junmin’s research team from the College of Chemistry and Environmental Engineering at Shenzhen University published a research paper titled "Enantioselective Synthesis of 2-Oxabicyclo[2.1.1]hexanes and Bicyclo[2.1.1]hexanes via Catalytic Asymmetric Intramolecular Photocycloadditions" in Nature Communications (Impact Factor: 15.7; JCR Q1, a TOP journal). Dr. Tian Dong, a postdoctoral fellow in the team, is the first author of the paper. Professor Zhang Junmin from Shenzhen University and Professor Jiang Zhiyong from Henan Normal University are the co-corresponding authors. Shenzhen University is the first completing affiliation.

Abstract: Due to the ubiquitous presence of ortho-disubstituted benzenes in pharmaceuticals and agrochemicals, the exploration of their bioisosteres incorporating three-dimensional bridged cyclic scaffolds has attracted significant interest among chemists in recent years. As a result, 4,5-disubstituted 2-oxabicyclo[2.1.1]hexanes have recently emerged as promising candidates owing to their enhanced pharmacological potency, improved metabolic stability, and, importantly, superior aqueous solubility. Herein, we report the first enantioselective synthesis of such compounds via an intramolecular [2 + 2] photocycloaddition enabled by cooperative photoinduced energy transfer (EnT) and chiral Brønsted acid catalysis. A broad range of valuable pyridine-functionalized 2-oxabicyclo[2.1.1]hexane derivatives were obtained in high yields with good to excellent enantioselectivity and diastereoselectivity. In addition, this transition metal-free approach offers an efficient and modular route to pyridine-based bicyclo[2.1.1]hexanes, which contain a significant yet challenging structural motif—pyridine rings directly attached to the bridgehead carbon atoms. Mechanistic studies demonstrate that the photosensitizer and low reaction temperature are crucial for rendering the chiral catalyst indispensable in facilitating substrate activation to the triplet state, thereby enabling enantiocontrol in this highly reactive transformation.

Link to the paper:https://www.nature.com/articles/s41467-026-71590-x
Professor Zhang Junmin’s research team from the College of Chemistry and Environmental Engineering at Shenzhen University published a research paper titled "Pd1Aul/Graphene: Highly Efficient Bimetallic Single-Atom Catalysts for C-C Coupling from C-B Bond Activation" in ACS Nano (Impact Factor: 16.0; JCR Q1, a TOP journal). Researcher Zhang Yujun from Shenzhen University and Zhejiang University and PhD student Teng Hui from Shenzhen University are the co-first authors of the paper. Professor Zhang Junmin from Shenzhen University, along with Professor Sun Fei and Professor Yu Xinghua from Zhejiang University, are the co-corresponding authors. Shenzhen University is the first completing affiliation.

Abstract: Bimetallic single-atom catalysts (SACs) represent an emerging paradigm beyond single-atom catalysts for enabling synergistic catalysis. The homocoupling of arylboronic acids via C–B bond activation to form C–C bonds has been demonstrated to be entirely inert with Pd or Au SACs. This study achieves a breakthrough by developing a bimetallic single-atom catalyst (Pd1Au1/graphene), which delivers outstanding performance with a TON up to 19,192 and cycling stability (>90% yield after 7 cycles). Through comprehensive experiments and theoretical calculations, we have identified a structure of PdN4-AuN4/Graphene, where adjacent Pd and Au atoms form a synergistic active center. This promotes simultaneous aromatic boronic acid substrate activation and significantly reduces the reaction energy barrier of the oxidative addition step, thereby enabling a pathway inaccessible to isolated single atoms. This work not only provides an efficient catalyst for organic synthesis but also validates the fundamental principle of synergistic catalysis in bimetallic single-atom catalysts, offering a universal design strategy for addressing challenging transformations.

Link to the paper:https://pubs.acs.org/doi/full/10.1021/acsnano.6c01027
Professor Zhang Junmin’s research team from the College of Chemistry and Environmental Engineering at Shenzhen University published a research paper titled "Photocatalytic C(sp²)–O Bond Cleavage and Dearomatization of Anisoles toward Epoxyquinols/Para�Quinols" in Organic Letters (Impact Factor: 5.0; JCR Q1, a TOP journal). Associate Researcher Xi Longyi and master student Liao Bilin from the team are the co�first authors of the paper. Professor Zhang Junmin from Shenzhen University is the sole corresponding author, and Shenzhen University is the sole completing affiliation.

Abstract: Cleavage of C(sp²)-O bonds in anisoles typically requires transition metal catalysis or harsh reaction conditions. Herein, we report a mild and metal-free photocatalytic strategy for the direct conversion of anisole derivatives into epoxyquinols and para-quinols. This method uses in-situ generated singlet oxygen and water as a nucleophile to control divergent pathways. The transformation exhibits broad substrate scope, excellent functional group tolerance, and can be applied to the late-stage functionalization of complex natural products. This work provides a sustainable alternative to traditional transition-metal-catalyzed C(sp²)-O functionalization and expands photoredox catalysis in dearomative synthesis.

Link to the paper:https://pubs.acs.org/doi/10.1021/acs.orglett.6c01258
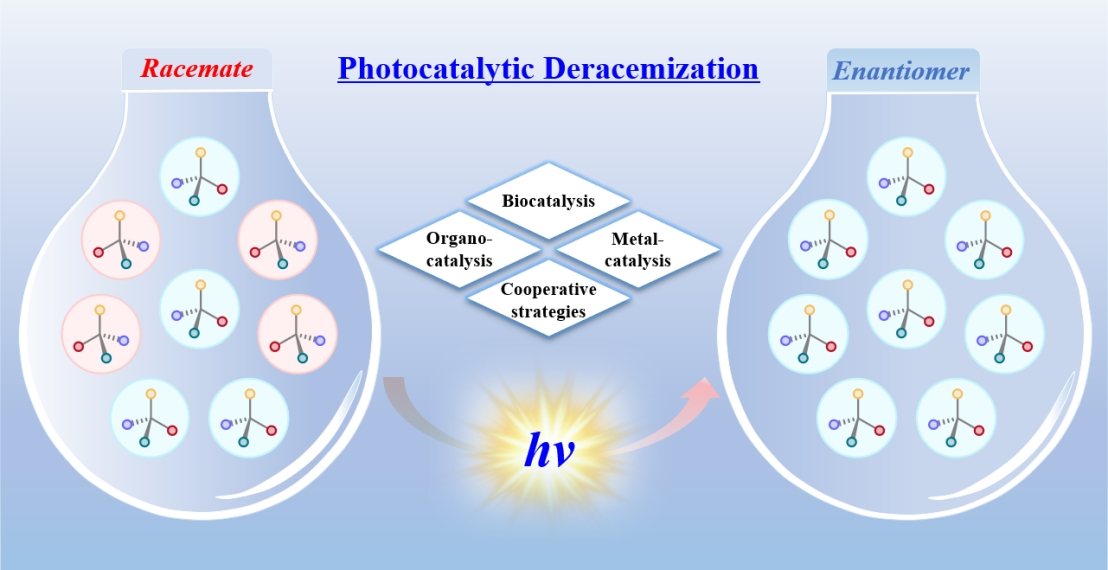
Professor Zhang Junmin’s research team from the College of Chemistry and Environmental Engineering at Shenzhen University published a review paper titled "Advances in photocatalytic deracemization reactions" in Science China Chemistry (Impact Factor: 9.7; JCR Q1, a TOP journal). Dr. Tang Yi, a postdoctoral fellow in the team, is the first author of the paper. Professor Zhang Junmin from Shenzhen University and Professor Tan Bin from Southern University of Science and Technology are the co�corresponding authors. Shenzhen University is the first completing affiliation.

Abstract: Direct deracemization of racemates presents an ideal access to acquire highly enantioenriched molecules. Research in photocatalytic deracemization, since its first demonstration in 2018, has been advancing steadily and witnessed explosive growth in 2025, becoming a more diverse and active field. This review comprehensively summarizes recent advances in photocatalytic deracemization by categorizing the catalytic systems employed, including organocatalysis, metal catalysis, biocatalysis, or cooperative strategies, with a focus on their evolution and mechanisms. The diversification of catalytic systems has advanced the understanding of photochemical stereocontrol and offered useful guidance for the development of novel catalysts and the optimization of reaction systems. Moreover, these advances have laid a solid foundation for the preparation of valuable chiral compounds, enabling their broader application in pharmaceutical development and materials science.
Link to the paper:https://www.sciengine.com/SCC/doi/10.1007/s11426-026-3489-2
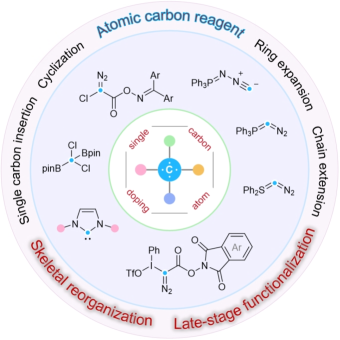
Professor Zhang Junmin’s research team from the College of Chemistry and Environmental Engineering at Shenzhen University published a review paper titled "Precisely Skeletal Reorganization via Single Carbon Atom Insertion" in Chinese Journal of Chemistry (Impact Factor: 5.5; JCR Q1, a TOP journal). Dr. Da Bingchao, a postdoctoral fellow in the team, is the first author of the paper. Professor Zhang Junmin from Shenzhen University and Professor Tan Bin from Southern University of Science and Technology are the co-corresponding authors. Shenzhen University is the first completing affiliation.

Abstract: Single carbon atom insertion (SCAI) has rapidly evolved from a specialized synthetic curiosity into a sophisticated and indispensable paradigm for modern molecular scaffold editing. By leveraging simple, readily available precursors, this versatile strategy provides a streamlined and remarkably efficient avenue for constructing complex molecular architectures through a variety of transformative pathways, including cyclization, ring expansion, chain extension, and direct atomic embedding. Fundamentally, SCAI offers a logically distinct alternative to conventional multi-step synthesis, which often relies on the tedious, bond-by-bond assembly of a framework. Instead, it empowers synthetic chemists to perform precise skeletal reorganization on existing scaffolds, significantly enriching the chemical toolbox and enabling direct access to structurally diverse core frameworks that are ubiquitous in pharmaceuticals and bioactive natural products. The field has been further propelled by the design of innovative, stable, and scalable atomic carbon precursors that circumvent the hazards associated with traditional, highly reactive reagents. The successful integration of these precursors with emerging synthetic technologies, such as photocatalysis and electrochemistry, has allowed various SCAI transformations to proceed under exceptionally mild and sustainable conditions. Crucially, many of these modern methods bypass the requirement for costly or toxic transition metal catalysts, which not only enhances reagent controllability and substrate generality but also drastically improves the overall reaction efficiency. These advancements have fundamentally expanded the boundaries of chemical application, moving the technique from theoretical exploration to practical utility in high-stakes synthesis. Today, by enabling the direct incorporation of a single carbon atom into established molecular frameworks, SCAI has become a vital instrument for late-stage functionalization and the strategic expansion of complex ring systems. This capability is particularly invaluable in the synthesis of natural products and drug discovery, where maintaining high structural fidelity while exploring new chemical space is paramount. As researchers continue to refine these methods, SCAI stands as a transformative approach that maximizes atom economy and provides an elegant solution to some of the most challenging structural modifications in organic chemistry.
Link to the paper:https://onlinelibrary.wiley.com/doi/abs/10.1002/cjoc.70514
